The world around you is constantly changing. Whether it is the passing of the seasons, a cool breeze or ice blocks melting in a drink, change is occurring. Most of these changes can be classified as physical or chemical changes depending on whether any new substances are produced.
Reference 1: Core Science Stage 4 Chapter 12: Chemical Changes
Reference 2: Pearson Science 8 NSW chapter 7: Physical and Chemical Change
Reference 1: Core Science Stage 4 Chapter 12: Chemical Changes
Reference 2: Pearson Science 8 NSW chapter 7: Physical and Chemical Change
|
Core Learning Outcomes
|
Homework and Informal Assessment
|
Task1: 1.6) Physical and Chemical Changes:
A physical change is when no new substance is formed. This can be when a substance changes size, or shape or state. Sometimes when we mix substances together like water and salt, the salt dissolves into the water. It is easy to get the salt back as it is just sitting in the water. This is an example of a physical change as no new substance has been formed. A change of state is when a substance changes from solid to a liquid or a gas. Think of water - ice, liquid water or steam. Read Core Science Stage 4 Chapter 12 Section 12.1) Time for a Change and list some examples.
A chemical change is when a new substance is formed and it is very difficult to reverse this change. There are a number of clues that indicate that a chemical reaction has occurred. They are:
A physical change is when no new substance is formed. This can be when a substance changes size, or shape or state. Sometimes when we mix substances together like water and salt, the salt dissolves into the water. It is easy to get the salt back as it is just sitting in the water. This is an example of a physical change as no new substance has been formed. A change of state is when a substance changes from solid to a liquid or a gas. Think of water - ice, liquid water or steam. Read Core Science Stage 4 Chapter 12 Section 12.1) Time for a Change and list some examples.
A chemical change is when a new substance is formed and it is very difficult to reverse this change. There are a number of clues that indicate that a chemical reaction has occurred. They are:
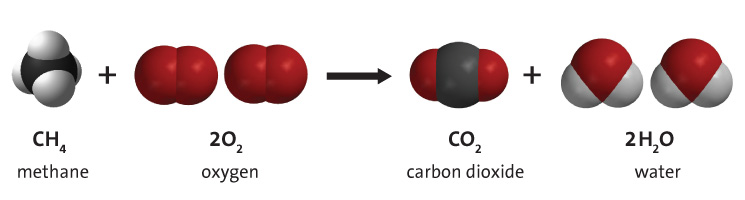
When a chemical change occurs, we can write a chemical word equation. The substances that we start with are called REACTANTS and the substances that we end up with are called PRODUCTS. Here is an example of a chemical word equation for when natural gas (methane) burns on a stove to produce carbon dioxide and water vapour. Remember when substance is burning it is reaacting with oxygen.
1) Read Core Science Stage 4) Section 12.2) Describing Chemical Changes especially paying attention to:
2) Complete the Activities at the end of section 12.2) Q 2, 3, 4 and 7
- reactants and products
- how to write word equations
2) Complete the Activities at the end of section 12.2) Q 2, 3, 4 and 7
|
Task 2: Video on Physical and Chemical Changes
Watch this fun animated video "Physical and Chemical changes of Matter" to increase your understanding |
Task 6: Model Activity
|
Additional Resources:
1. Video on Physical and Chemical Changes 2. A very good Website "Physical and Chemical Changes" |
|
Revision for Topic Test
1) Core Science Stage 4 - Looking Back on pages 299 - 300 Questions