4.1) The Structure of the Atom
All Chemistry involves an understanding of how the atom works and how atoms react with each other.
The information below provides an in-depth revision of year 9
Review of Definitions
A) Elements
|
An Element is a substance that is made up of only one type of atom. Eg pure gold consists of only gold atoms OR hydrogen gas consists of only hydrogen atoms.
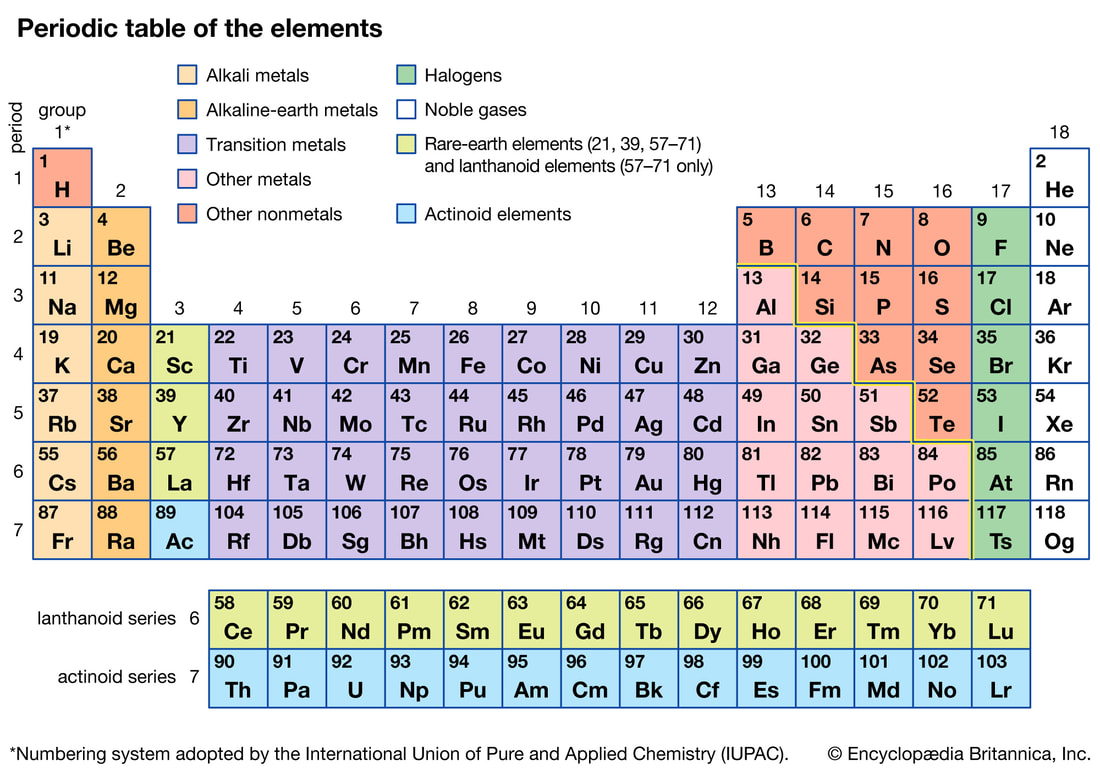
There are 98 naturally-occurring elements and 20 man-made elements which have been specially arranged into a table known as the Periodic Table. Atoms of the same type can also be arranged differently to give different versions of an element eg Carbon can have its atoms arranged differently to form soot (or charcoal) or diamond or graphite. Each element has a unique set of properties which allows us to identify that element. |
B) Compounds
Most substances that you encounter in everyday life are made up of more than one type of atom. These are called compounds. Compounds are formed from at least two or more different types of atoms that are chemically bonded together. Like elements, every compound has a unique set of characteristics. These properties are usually very different to the properties of the elements that make up the compound.
Think of table salt which is made of sodium and chlorine, two toxic elements that chemically combine to give an edible solid compound
Think of table salt which is made of sodium and chlorine, two toxic elements that chemically combine to give an edible solid compound
C) Pure Substances and Mixtures
|
Pure Substances contain only one type of atom or compound. For example pure gold only contains gold particles AND pure water contains only water molecules.
Mixtures are when the elements and/or compounds are not chemically bonded together. For example air is a mixture of the elements nitrogen and oxygen with the compounds carbon dioxide, carbon monoxide and many others. Unlike elements and compounds, you cannot write a chemical formula for mixtures as they are not chemically joined together |
D) Common Elements and Compounds
|
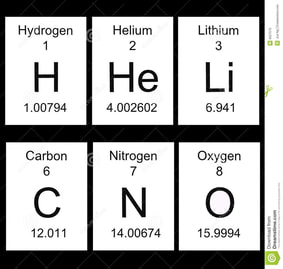
Here is a list of the first 20 elements and some of the common compounds that you should memorise
|
E) Atoms and Molecules
Atoms are the simplest form of matter. An atom consists of a single particle of matter. An example a single gold atom.
Molecules are made up of two or more atoms joined together by chemical bonds. In molecules, the atoms can be all the same or they may be different. For example hydrogen gas or carbon dioxide
There are three ways of correctly describing matter
1) the atom of an element - A gold atom can be described as an atom of an element.
2) the molecule of an element - Hydrogen gas is described as a molecule of an element. It is a molecule as the hydrogens are chemically bonded together and it is an element as it contains only hydrogen atoms.
3) the molecule of a compound - Carbon dioxide can be described as a molecule of a compound as it is molecule as the carbon and two oxygens are chemically bonded together and it is a compound as it contains two different types atoms.
Molecules are made up of two or more atoms joined together by chemical bonds. In molecules, the atoms can be all the same or they may be different. For example hydrogen gas or carbon dioxide
There are three ways of correctly describing matter
1) the atom of an element - A gold atom can be described as an atom of an element.
2) the molecule of an element - Hydrogen gas is described as a molecule of an element. It is a molecule as the hydrogens are chemically bonded together and it is an element as it contains only hydrogen atoms.
3) the molecule of a compound - Carbon dioxide can be described as a molecule of a compound as it is molecule as the carbon and two oxygens are chemically bonded together and it is a compound as it contains two different types atoms.
Review how to draw atoms using the Periodic table
To draw an atom, we need to work out the number of protons, neutrons and electrons using the Periodic Table. Read through the information provided and make suitable notes.
A) Information about Atoms
A) Information about Atoms
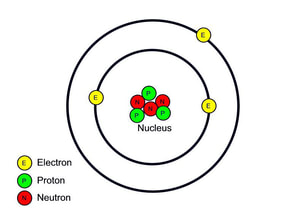
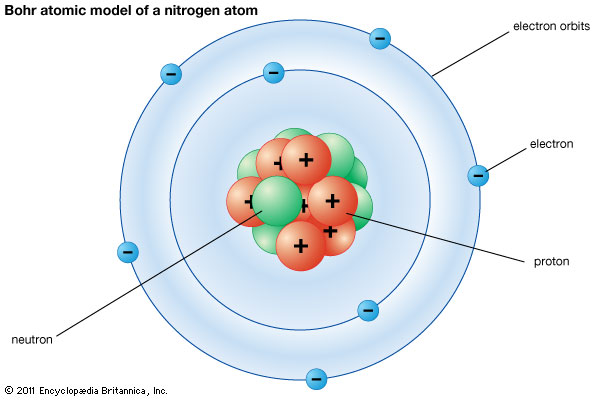
- Atoms consist of PROTONS (p), NEUTRONS (n) and ELECTRONS (e).
- Protons and Neutrons are found in the NUCLEUS
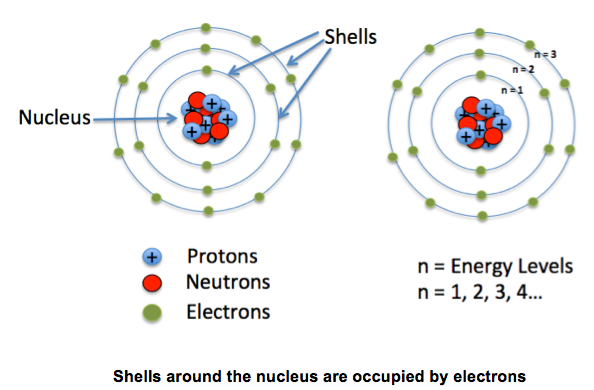
- Electrons orbit around the nucleus in shells or orbits or energy levels
- Protons are positively charged, neutrons are neutral and electrons are negatively charged.
B) Information about the Number of Protons and the Number of Electrons:
- The number of protons is determined from the periodic table. It is the smaller of the two numbers given for an element. This number has a special name - The Atomic Number.
- The number of electrons for an atom is the same as the number of protons.
- Example 1: This is the LITHIUM atom. It is the THIRD element on the Periodic Table and this means that it has THREE PROTONS. It's atomic number is also 3. It also has three electrons.
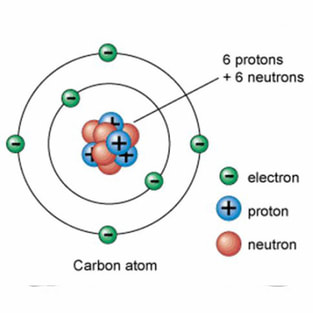
Example 2: This is the CARBON atom. It is the SIXTH element on the Periodic Table and has SIX PROTONS and SIX ELECTRONS.
C) Information about the Number of Neutrons:
- The number of neutrons is found by using both numbers given in the periodic table for that element. The larger number is called the ATOMIC MASS.
- The number of neutrons is found by
|
D) Information about Electron Shells
The Electrons are nearly 2000 times smaller than protons and neutrons. These electrons fly a long way away from around the nucleus in orbits. Sometimes the orbits are called shells or energy levels. Orbit 1 = 2 electrons Orbit 2 = 8 electrons Orbit 3 = 8 electrons Orbit 4 = 8 electrons |
Important Skill) Why do we need Scientific Models?
In science, a model is a representation of an idea, an object or even a process or a system that is used to describe and explain phenomena that cannot be experienced directly. Models are central to what scientists do, both in their research as well as when communicating their explanations. Models are a mentally visual way of linking theory with experiment, and they guide research by being simplified representations of an imagined reality that enable predictions to be developed and tested by experiment.
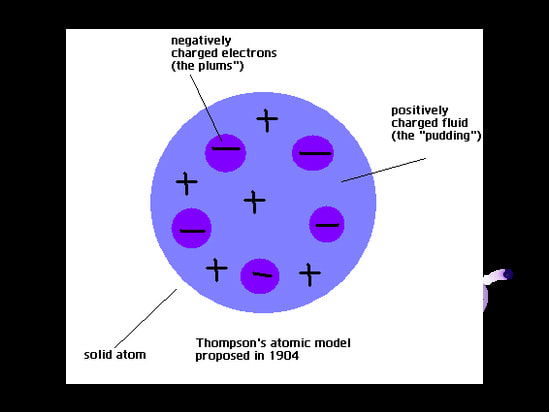
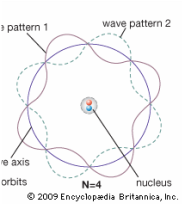
Models have always been important in science and continue to be used to test hypotheses and predict information. Often they are not accurate because the scientists may not have all the data. It is important that scientists test their models and be willing to improve them as new data comes to light. Think about our Model of the Atom. In 1897, J J Thomson proposed the "Plum Pudding" model of the atom after he discovered the electron. Then from 1912 to 1913, Rutherford and Bohr further developed the model of the atom after proton and neutron were discovered. Now we have the Quantum Model of the Atom which was proposed in the 1930's. In this model, the electrons do not exist as particles of matter but as waves of matter that extend around the atom.
In science, a model is a representation of an idea, an object or even a process or a system that is used to describe and explain phenomena that cannot be experienced directly. Models are central to what scientists do, both in their research as well as when communicating their explanations. Models are a mentally visual way of linking theory with experiment, and they guide research by being simplified representations of an imagined reality that enable predictions to be developed and tested by experiment.
Models have always been important in science and continue to be used to test hypotheses and predict information. Often they are not accurate because the scientists may not have all the data. It is important that scientists test their models and be willing to improve them as new data comes to light. Think about our Model of the Atom. In 1897, J J Thomson proposed the "Plum Pudding" model of the atom after he discovered the electron. Then from 1912 to 1913, Rutherford and Bohr further developed the model of the atom after proton and neutron were discovered. Now we have the Quantum Model of the Atom which was proposed in the 1930's. In this model, the electrons do not exist as particles of matter but as waves of matter that extend around the atom.
|
Extension Activity 1) - Construct a 3D Model of an Atom
1. Research the structure of an atom and draw in your books. All components must be labelled. 2. Write down the meaning of "atomic number", "atomic mass" 3. Draw diagrams of the following three atoms, showing the correct number of protons, electrons, neutrons. The electrons must also be in their correct energy level or orbit. Find out any interesting information on your atom using WebElements website. a) Boron b) Nitrogen c) Neon 4. Create a model of your atom from the equipment provided. You will present your model and help the class to understand the structure of a model. |
Extension Activity 2) The Historical Development of Atomic Theory
Create a Timeline of the Development of the Atomic Model
a) First you will conduct research on your group's scientist
Group 1: Democritus Group 5: Ernest Rutherford
Group 2: Lavoisier Group 6: Bohr
Group 3: Dalton Group 7: Modern Quantum Theory
Group 4: JJ Thompson
You must find the following information about your scientist
* Full Name
* Date of Important Discovery or Birth Date
* Information on his contribution to atomic theory
* Other interesting information or diagrams
b) Share information to the class
c) Construct a SCALED timeline where 1 cm = 100 cm
a) First you will conduct research on your group's scientist
Group 1: Democritus Group 5: Ernest Rutherford
Group 2: Lavoisier Group 6: Bohr
Group 3: Dalton Group 7: Modern Quantum Theory
Group 4: JJ Thompson
You must find the following information about your scientist
* Full Name
* Date of Important Discovery or Birth Date
* Information on his contribution to atomic theory
* Other interesting information or diagrams
b) Share information to the class
c) Construct a SCALED timeline where 1 cm = 100 cm
|
|
|
|
|
|
|
|