4.3) Radioactivity
Interest - Here are some interesting radioactive products that were sold.

Interest - Today Uranium Glass can be purchased on ebay and many antique stores around Australia. Read the article "These people love to collect Radioactive Glass" so you can answer the following questions:
a) What is Uranium Glass?
b) Is it dangerous?
c) How much Uranium is in it?
d) It usually glows a greeny colour when ultraviolet light is shone onto it. How does Uranium produce this green glow?
|
The Discovery of Radioactivity - Marie and Pierre Curie came to work with Becquerel to investigating the invisible rays that his uranium samples had produced. They found that the rays produced by uranium ores caused the air particles around them to become ionised. They referred to the rate at which the ionisation occurred as activity and the substances that produced this activity as being radioactive. Read about their discovery at the "Awesome Stories" webpage.
In the 1900's, people were very excited about radium - a radioactive substance. Not much was understood about radioactivity so people started using it for all different things. |
Task 1) Write these notes about Isotopes and Radioisotopes
If you change the number of PROTONS in the nucleus then you will create a different element. For example Helium has 2 protons and if you add one more proton then helium will change to lithium.
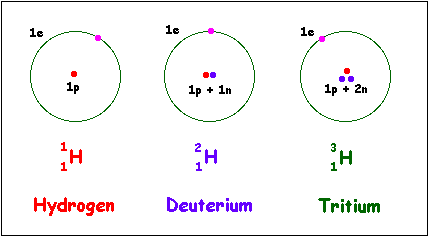
However you can change the number of neutrons and this will create different ISOTOPES of the element. An example for hydrogen there are three isotopes - Hydrogen-1, Hydrogen-2 and hydrogen-3. Each isotope of hydrogen has 1 proton but they differ in the number of neutrons
If you change the number of PROTONS in the nucleus then you will create a different element. For example Helium has 2 protons and if you add one more proton then helium will change to lithium.
However you can change the number of neutrons and this will create different ISOTOPES of the element. An example for hydrogen there are three isotopes - Hydrogen-1, Hydrogen-2 and hydrogen-3. Each isotope of hydrogen has 1 proton but they differ in the number of neutrons
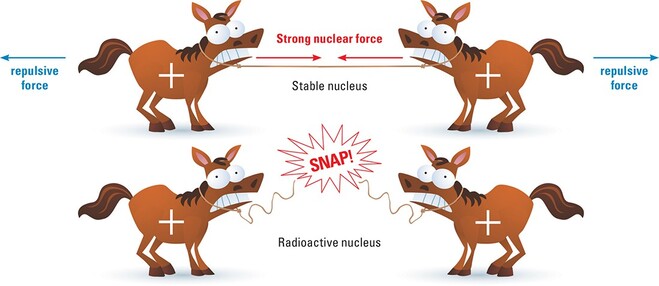
The protons and neutrons of the nucleus are held tightly together by something called the STRONG NUCLEAR FORCE. Without the strong nuclear force, the repulsive electrostatic force between the positively charged protons would tear the nucleus apart. In most isotopes, the repulsive electrostatic force and the strong nuclear force equal and the nucleus remains intact or STABLE.
In some isotopes, however the extra neutrons in the nucleus make it much harder for the strong nuclear force to keep the nucleus together and the nucleus breaks apart or DECAYS. When an unstable nucleus breaks up, energy in the form of radiation is released and smaller, more stable elements are formed. As a result, isotopes whose nuclei have a tendency to decay are said to be RADIOACTIVE.
In some isotopes, however the extra neutrons in the nucleus make it much harder for the strong nuclear force to keep the nucleus together and the nucleus breaks apart or DECAYS. When an unstable nucleus breaks up, energy in the form of radiation is released and smaller, more stable elements are formed. As a result, isotopes whose nuclei have a tendency to decay are said to be RADIOACTIVE.
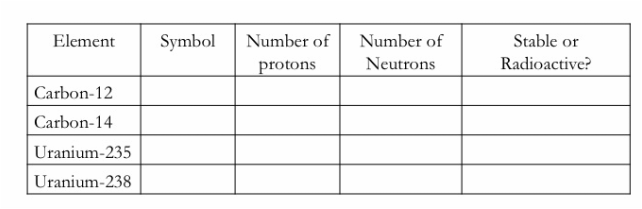
Below is a table of different elements that are also found in the radioactive form through the addition of neutrons in the nucleus.
|
Task 2) Write notes about Alpha Particles, Beta Particles and Gamma Radiation
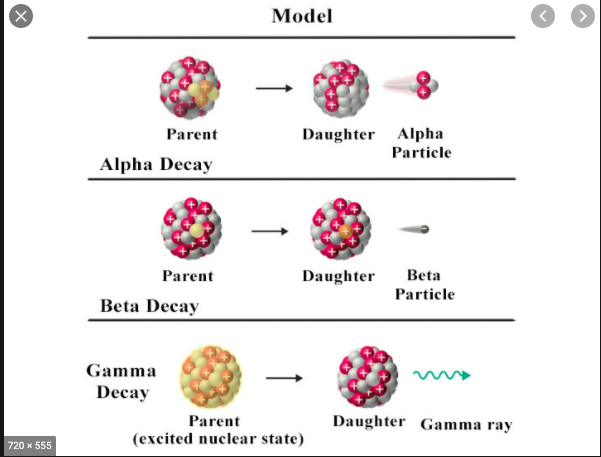
In this task, you will learn about the three different types of emissions from a radioactive particle. What are they and what are their properties. When radioactive substances, like uranium-238 and carbon-14, decay then their nuclei break apart releasing particles and energy. These particles can be alpha or beta particles and the energy released is gamma radiation as the diagram opposite shows. |
|
Watch this youtube video and fill in the worksheet provided.
|
Interesting Demonstration of the different types of Radioactive Decay
|
|
Extension Task - Writing Radioactive Decay Equations
a) Watch the video b) Complete the worksheet |
|
4.4) Half Life

Task 1) Conduct an experiment to learn about Half-Life of Radioactive Substances
Let's see what we can learn about radioactivity using M&M's in the practical "Simulating Radioactive Decay"
NOTE: An important skill in this practical is drawing the graph. Make sure that you use a ruler and a pencil as well as labelling the axes correctly.
Let's see what we can learn about radioactivity using M&M's in the practical "Simulating Radioactive Decay"
NOTE: An important skill in this practical is drawing the graph. Make sure that you use a ruler and a pencil as well as labelling the axes correctly.
|
Watch this youtube clip to understand more about your experiment and radioactive decay.
|
|
Task 2) Half-Life and Radioactive Decay
|
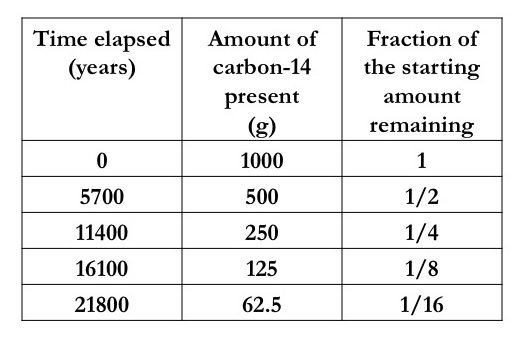
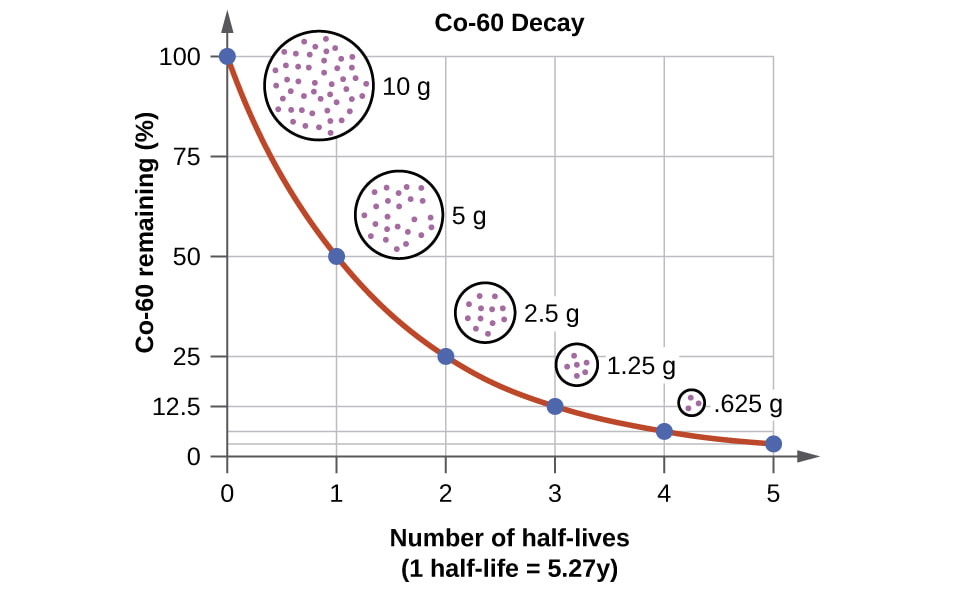
The rate at which radioisotopes decay is described by its half-life — the time you would need to wait for half of the amount of the radioactive material to decay.
For example, carbon-14 has a half-life of 5700 years. If you had a 1 kg lump of carbon-14, after 5700 years had elapsed, only 500 g would still be carbon-14. The other 500 g would have decayed to form the stable isotope nitrogen-14 (we say that nitrogen-14 is the daughter isotope of carbon-14). After 11 400 years, you would have only 250 g of carbon-14 and after 16 100 years only 125 g of the carbon-14 would be left. The table on the right shows the decay of carbon-14. |
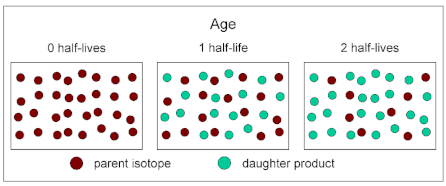
We can also represent radioactive decay in the form of a particle diagram or as a graph.
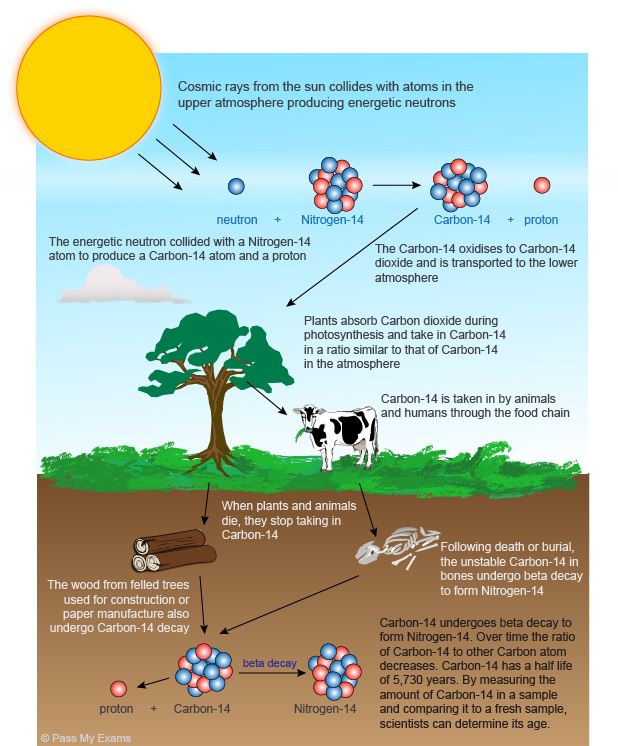
Radiocarbon Dating is a particularly useful tool when scientists are attempting to determine the age of a fossil. Read through this diagram about radiocarbon dating and answer the questions below. You will need to use the video on the Scientific American website called "How does Radiocarbon Dating work?" to answer some of the questions.
|
Questions
1) How do plants get carbon-14? 2) How do animals get carbon-14? 3) Why does this stop when the organism dies? 4) How many protons and neutrons does carbon-12 have? 5) What is the difference between carbon-12 and carbon-14? 6) What does carbon-14 decay into? 7) How do we use carbon-12 and carbon-14 to measure when a fossil died? 8) What is the maximum age that a fossil can be dated using carbon-14? 9) Why can scientists only date fossils up to 60,000 year old? 10) What other method can be employed to date a fossil if it is older? |
4.5) Benefits and Limitations of using Radioactive Isotopes and Nuclear Energy
Question: What are some of the uses of Radioisotopes and Nuclear Energy? Complete the worksheet
|
"Radioactivity is often referred to as a double-edged sword. There are many benefits that can be gained from radioactive isotopes however there are also various problems that have arisen. Overall it is often believed that Radioactivity has been a benefit for society."
1) Watch the video "Nuclear Power: Risks and Benefits" and complete the Quiz 2) Read through this document and then ASSESS this statement using evidence provided from this resource and any other VALID SCIENTIFIC resource that you find that supports your conclusion. Your extended response should be at least 3/4 pg if typed in Garamond 12 Font. |
|
|
Inside Chernobyl
Chernobyl Catastrophe) was a catastrophic nuclear accident that occurred on 26 April 1986 at the Chernobyl Nuclear Power Plant in Ukraine (then officially Ukrainian SSR), which was under the direct jurisdiction of the central authorities of the Soviet Union. An explosion and fire released large quantities of radioactive particles into the atmosphere, which spread over much of Western USSR and Europe. The Chernobyl disaster is widely considered to have been the worst nuclear power plant accident in history, and is one of only two classified as a level 7 event on the International Nuclear Event Scale (the other being the Fukushima Daiichi nuclear disaster in 2011) The battle to contain the contamination and avert a greater catastrophe ultimately involved over 500,000 workers and cost an estimated 18 billion rubles. The official Soviet casualty count of 31 deaths has been disputed, and long-term effects such as cancers and deformities are still being accounted for. |
|
|
|
Seconds from Disaster - The Meltdown at Fukushima
The most recent nuclear disaster - The Fukushima Nuclear Disaster that was triggered when a tsunami crippled the nuclear plant's generators. |