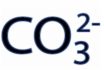Essential QuestionHow do Acids and Bases affect our lives???
1.17) What is an Acid and a Base? 1.18) The Reaction of Acids and Metals 1.19) The Reaction of Acids and Carbonates 1.20) Neutralisation |
The Core Learning Intentions
After completing the activities on this page you should be able to
|
|
1.17) What is an Acid and a Base?
Read SF3 Unit 3.4) Acids and Bases pg 88 - 93 and then demonstrate your understanding by completing the worksheet below.
| |||||||
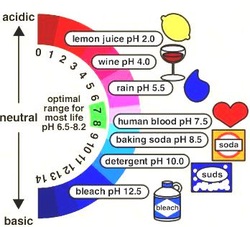
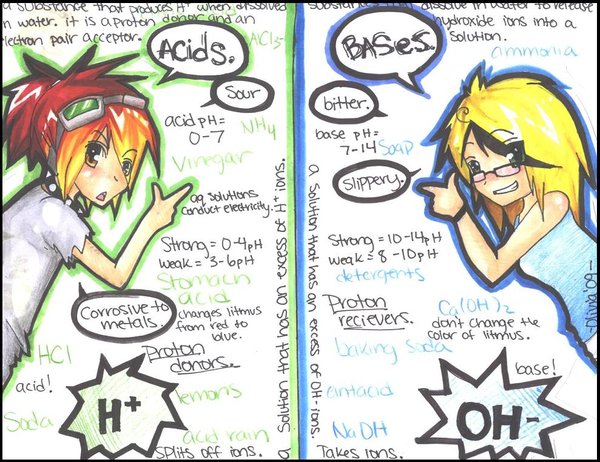
Acids are corrosive substances. That means that they react with solid substances including metals, marble and even the enamel of your teeth, effectively ‘eating’ them away. When strong acids such as the sulfuric acid used in car batteries come into contact with the skin, they break down the proteins and fats in the living tissues. This reaction is exothermic so the area heats up dramatically. At best, the result is an acid burn that is slow to heal.
However, not all acids are strong acids. The weaker acids in ant and bee stings may cause pain but there is little lasting damage to the tissues around the sting site. Others, like the acids in citrus fruits and vinegar, are safe — even pleasant — to taste. Acids such as these are routinely added to food to give it the distinctive sour taste associated with acids — in fact, the word ‘acid’ comes from the Latin word acidus meaning sour. Acids can also be used to preserve food or to react with other substances in food to produce carbon dioxide gas which makes foods like sponge cake light and fluffy.
Name the acid in each of the images below:
However, not all acids are strong acids. The weaker acids in ant and bee stings may cause pain but there is little lasting damage to the tissues around the sting site. Others, like the acids in citrus fruits and vinegar, are safe — even pleasant — to taste. Acids such as these are routinely added to food to give it the distinctive sour taste associated with acids — in fact, the word ‘acid’ comes from the Latin word acidus meaning sour. Acids can also be used to preserve food or to react with other substances in food to produce carbon dioxide gas which makes foods like sponge cake light and fluffy.
Name the acid in each of the images below:
Acids you must remember - hydrochloric acid, nitric acid, sulfuric acid,
Other acids - acetic acid, carbonic acid, citric acid, formic acid, lactic acid,
Other acids - acetic acid, carbonic acid, citric acid, formic acid, lactic acid,
Bases have a bitter taste and feel slippery or soapy to touch. Some bases are very corrosive, especially caustic soda which will break down fat, hair and vegetable matter. Other bases are used in shampoos, toothpaste, and cleaning agents like dishwashing liquid and cloudy ammonia. Bases that can be dissolved in water are called alkalis.
Name the bases in the substances below:
Name the bases in the substances below:
Bases you must remember - ammonia, calcium carbonate, sodium hydroxide
Other Bases - sodium bicarbonate
Other Bases - sodium bicarbonate
1.18) The Reaction of Acids and Metals
Acid + Metal ----> Salt + Hydrogen
What is a Salt???? A Salt is an ionic compound that is formed when the H+ ion from an acid is replaced with a positive ion!!!!
An Example:
Acid + Metal ----> Salt + Hydrogen
What is a Salt???? A Salt is an ionic compound that is formed when the H+ ion from an acid is replaced with a positive ion!!!!
An Example:
|
Practical - You will be working with your class group to complete a practical on the reaction of acids and metals. You will need to go to SF4 pg 99 and read through the instructions so you are ready for the practical. You will be given a hard copy in class. Ensure you answer all questions on the document. |

1.19) The Reaction of Acids and Carbonates
Now you are going to learn another common chemical reaction – Acids and Carbonates.
Acid + Carbonate -----> Salt + Water + Carbon dioxide
So what is a carbonate? Carbonate has a valency of -2 and the chemical formula is:
Now you are going to learn another common chemical reaction – Acids and Carbonates.
Acid + Carbonate -----> Salt + Water + Carbon dioxide
So what is a carbonate? Carbonate has a valency of -2 and the chemical formula is:
Examples:
Practical: Reaction of Acids and Carbonates
You will be working with your class group to complete this practical "Reaction of Acids and Carbonates". You will need to download the file below and read through the instructions so you are ready for the practical. You will be given a hard copy in class.
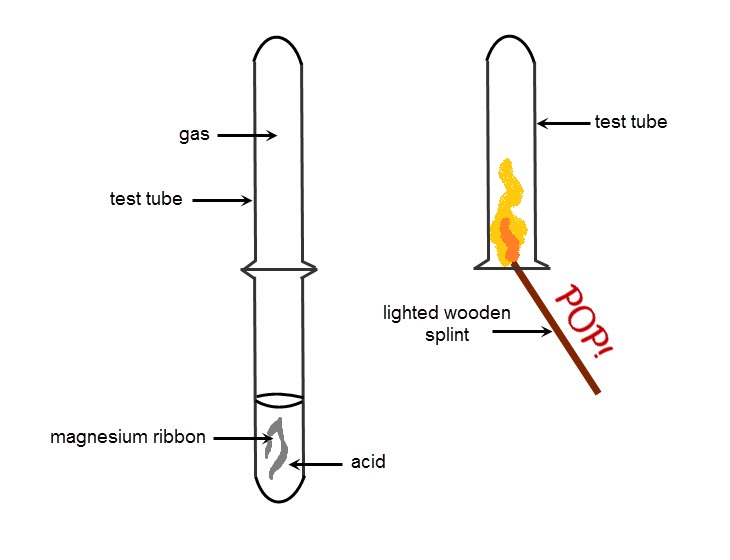
What are the TWO tests for carbon dioxide gas?
You will be working with your class group to complete this practical "Reaction of Acids and Carbonates". You will need to download the file below and read through the instructions so you are ready for the practical. You will be given a hard copy in class.
What are the TWO tests for carbon dioxide gas?
|
1.20) Neutralisation - The Reaction of Acids and Bases!
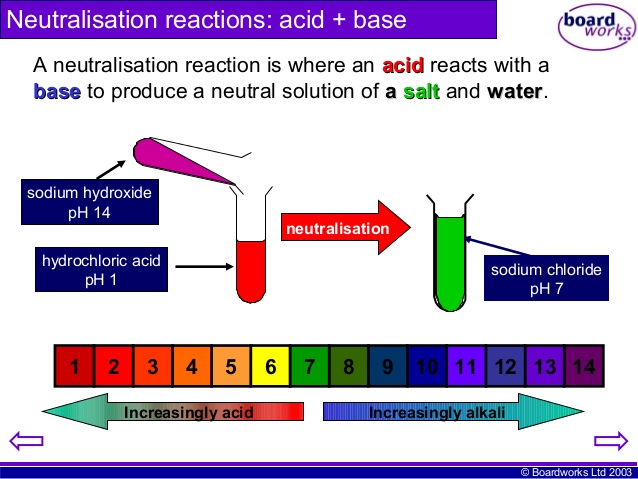
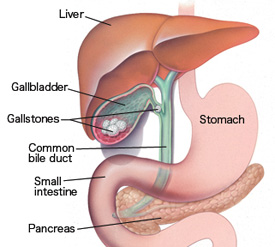
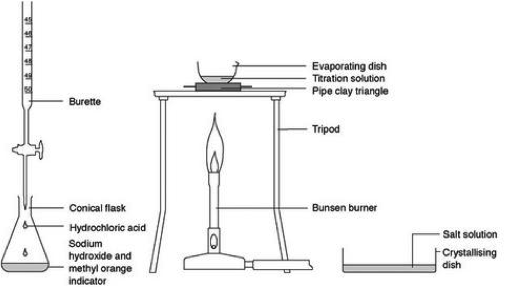
Neutralisation is when a BASE is added to an ACID in just the right quantites that the pH changes to 7 ie NEUTRAL. For example, the pain caused by the acidic sting of an ant can be relieved by washing the area with an alkali solution containing a weak base such as sodium bicarbonate (baking soda). Similarly, to stop a base from having an effect, an acid can be added. This is why the pain caused by the alkali in the sting of a wasp can be relieved by pouring a weak acid such as vinegar over the area. The Neutralisation Reaction is: ACID + BASE -----> SALT + WATER Extension: A base can be 1) Hydroxide eg NaOH OR 2) Oxide eg CaO Task 1) Read about Neutralisation in our everyday lives from CS5 section 7.4) Acids and Bases and answer the following questions in your exercise books: a) How do Antiacids help Indigestion? b) How does Self Raising Flour cause cakes to rise? c) How does Bile protect our intestines? Practical: The Reaction of Acids and Bases Titration is used to determine many amazing things in the world of chemistry. It is not easy and requires DEDICATION, PATIENCE and STEADY HANDS. Now that you have developed your practical skills, it is time to put them to the test. Find out if you can TITRATE??? |
|
Graphing Skills
|
|
|
Quick Revision of Acids and Bases
If you have forgotten your key concepts of Acids and Bases, simply click on the button and there is a quick revision for you to read. Further Revision of Acid and Bases
Read through this powerpoint for a thorough revision of acids, bases and indicators. |
|
|
Activity: Luxurious Homemade Moisturiser
| |||||||
|
Chocolate-Mint Homemade Lip Balm
| |||||||