1.3) Exploring Metals and Non-Metals
Mendeleev divided up the elements into three main groups - Metals, Non-Metals and Metalloids based on their properties. Lets see if we can conduct a practical to start working out the properties that he used to distinguish these groups.
Semi-Metals
These are elements that have both the properties of metals and non-metals. For example Silicon can be polished to shine like a metal but is a poor conductor of electricity and heat.
These are elements that have both the properties of metals and non-metals. For example Silicon can be polished to shine like a metal but is a poor conductor of electricity and heat.
|
Note: Malleable means that you can flattern the metal into shapes
AND Ductile means that you can draw the metals into wires |
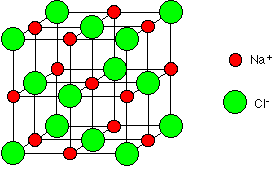
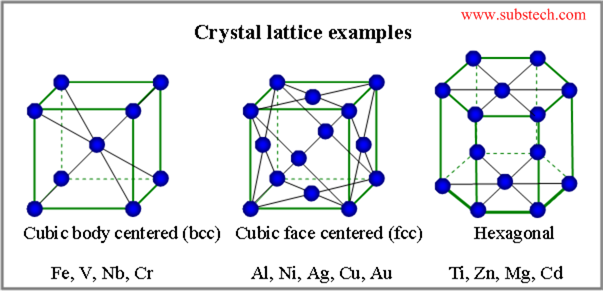
Lattice Structure means that the atoms bond very strongly to each other and hence they tend to be very hard and solids at room temperature. Two examples of lattices are table salt and metals.
|
|
To further develop your understanding of Metals and Non-Metals, watch this youtube video!
|
|