Topic 2) State of Change
Unit Summary
Everything on our planet and probably in the universe is made of elements shown in a periodic table. These elements can be classified based on their properties and can be rearranged into compounds which also possess properties which can impact on our lives in different ways. This topic introduces students to a couple of ways that the matter of which they are made can be organised, as well as how different materials can affect their lives. It also introduces students to the use of internationally agreed symbols which allows for easier communication between scientists.
Core Learning Outcomes
Knowledge and Understanding
Skills
Knowledge and Understanding
- recognize a physical change as one in which no new substances are formed and which is easily reversed
- recognise a chemical change as one in which new substances are formed and which is not easily reversed
- identify a chemical change by change in colour, production of gas, change in temperature, appearance of new substances
- identify the various changes of state - evaporation, condensation, melting, freezing
- describe the physical changes that occur during observations of evaporation, condensation, boiling, melting and freezing
- describe the behaviour of matter in terms of particles that are continuously moving and interacting
- describe expansion and contraction of materials in terms of a simple particle model
- relate an increase or decrease in the amount of energy possessed by particles to changes in particle movement
- relate properties of solids, liquids and gases to the particle model of matter
- explain density in terms of a simple particle model
- explain the changes in pressure of gases in terms of increases or decreases in frequency of particle collisions
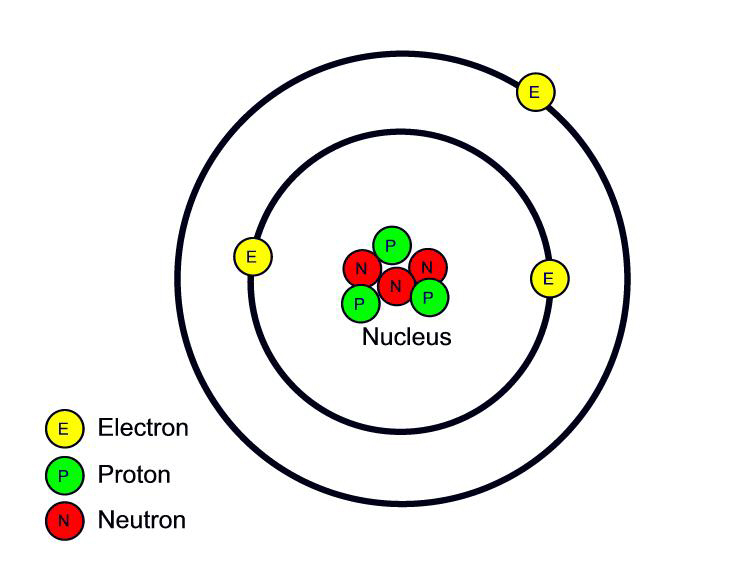
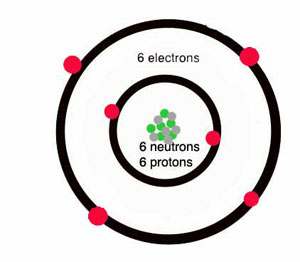
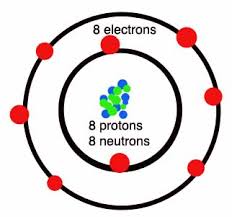
- distinguish between elements, compounds and mixtures in terms of the type and arrangement of particles
- classify elements as metals or non-metals according to common characteristics
- describe the different properties of metals and non metals
- describe the different properties of acids and bases
- Identify internationally recognised symbols for common elements
Skills
- perform a test for the presence of oxygen, carbon dioxide and hydrogen gas
- manipulate the mathematical formula for calculating density
- use a triple beam balance and electronic balance to correctly determine the weight of objects
Revision Sheet and Questions for Topic test on Monday 21st July 2014
1) Classify the following as elements, compounds or mixtures. The periodic table can be used to help you - lemonade, carbon, zinc, air, tap water, pure water, oxygen gas, sodium chloride, carbon dioxide, chlorine, shampoo and iron.
2) List the physical properties of metals and non-metals
3) If a substance is used to make bicycle frames, is light in weight but very strong and can be polished to a shiny finish. Is it a metal or a non-metal?
4) This substance has a low density, found in air and used by our bodies to make energy. Is it a metal or a non-metal?
5) Use the periodic table to identify the following elements:
1) Classify the following as elements, compounds or mixtures. The periodic table can be used to help you - lemonade, carbon, zinc, air, tap water, pure water, oxygen gas, sodium chloride, carbon dioxide, chlorine, shampoo and iron.
2) List the physical properties of metals and non-metals
3) If a substance is used to make bicycle frames, is light in weight but very strong and can be polished to a shiny finish. Is it a metal or a non-metal?
4) This substance has a low density, found in air and used by our bodies to make energy. Is it a metal or a non-metal?
5) Use the periodic table to identify the following elements:
| state_of_change_-_test_without_answers_v2.doc | |
| File Size: | 90 kb |
| File Type: | doc |
| state_of_change_-_test_with_answers_v2.doc | |
| File Size: | 122 kb |
| File Type: | doc |