4.2) Chemical Formula for Ionic Compounds
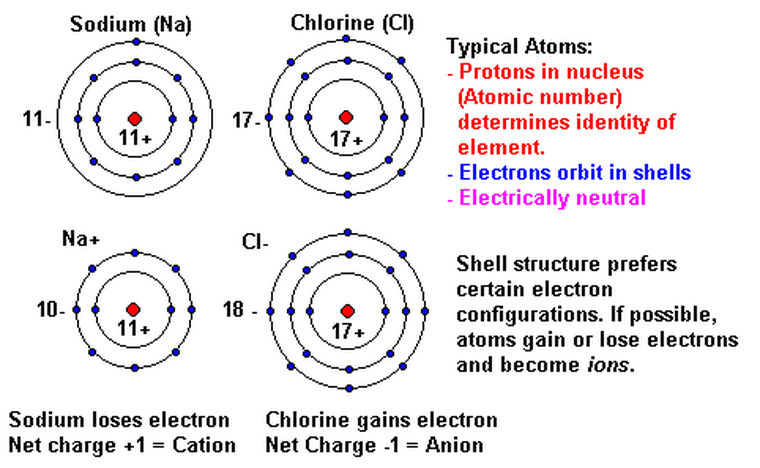
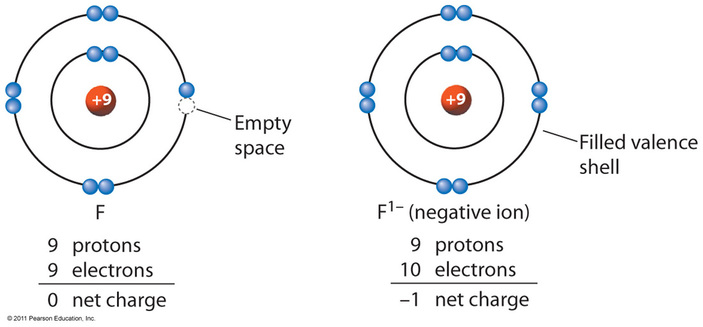
All atoms are neutral however they can lose or gain electrons to become ions. Metals form positive ions and Non-metals form negative ions. A metal ion and a non-metal ion come together to form ionic compounds. A common example of an ionic compounds is salt or sodium chloride
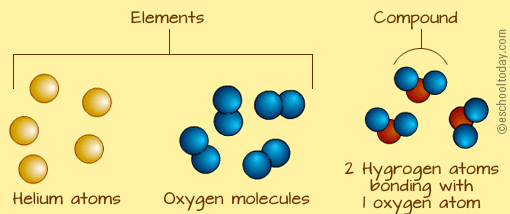
Task 1) Recall the difference between an atom, molecule, element and compound - don't do 2019
|
Research the definitions of atom, molecule, element and compound. Clarify the differences between these definitions with your group and/or teach Worksheet - Use this knowledge to complete the worksheet "Atoms, Molecules, Elements and Compounds" |
Task 2) Recall how atoms turn into positive or negative ions
|
Let's recall how neutral atoms turn into positive or negative ions by watching the youtube video called " Ion Formation"
|
|
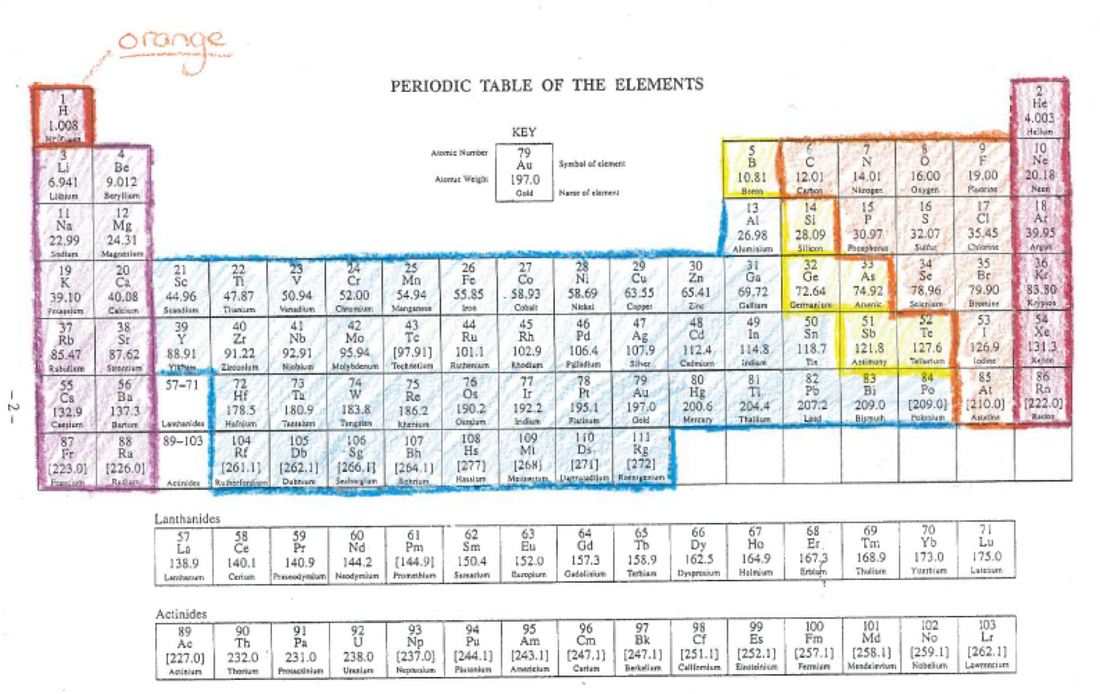
Task 3) Recall how to use the Periodic Table to determine Valencies of Ions
Column 1 = all these elements have one electron in their outermost shell and hence have a valency of +1
Column 2 = all these elements have two electrons in their outermost shell and hence have a valency of +2
Columns 3 - 12 = Most of theTransition metals usually have a valency of +2 but there are exceptions such as silver, Ag +1 and Fe can have +2 or +3
Column 13 = All all these elements have three electrons in their outermost shell and hence have a valency of +3
Column 14 = _______________________________________________________________________________________________________
Column 15 = _______________________________________________________________________________________________________
Column 16 = _______________________________________________________________________________________________________
Column 17 = _______________________________________________________________________________________________________
Column 18 = the Noble gases which have 8 electrons in their outermost shell and hence are full so unreactive
Column 2 = all these elements have two electrons in their outermost shell and hence have a valency of +2
Columns 3 - 12 = Most of theTransition metals usually have a valency of +2 but there are exceptions such as silver, Ag +1 and Fe can have +2 or +3
Column 13 = All all these elements have three electrons in their outermost shell and hence have a valency of +3
Column 14 = _______________________________________________________________________________________________________
Column 15 = _______________________________________________________________________________________________________
Column 16 = _______________________________________________________________________________________________________
Column 17 = _______________________________________________________________________________________________________
Column 18 = the Noble gases which have 8 electrons in their outermost shell and hence are full so unreactive
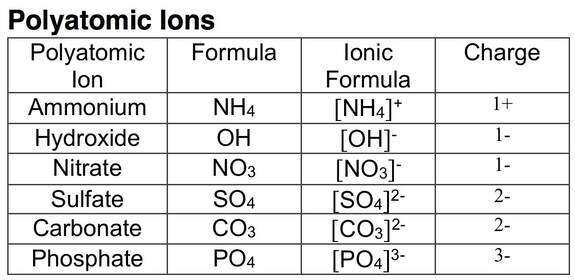
Task 4) Recall the Polyatomic Ions
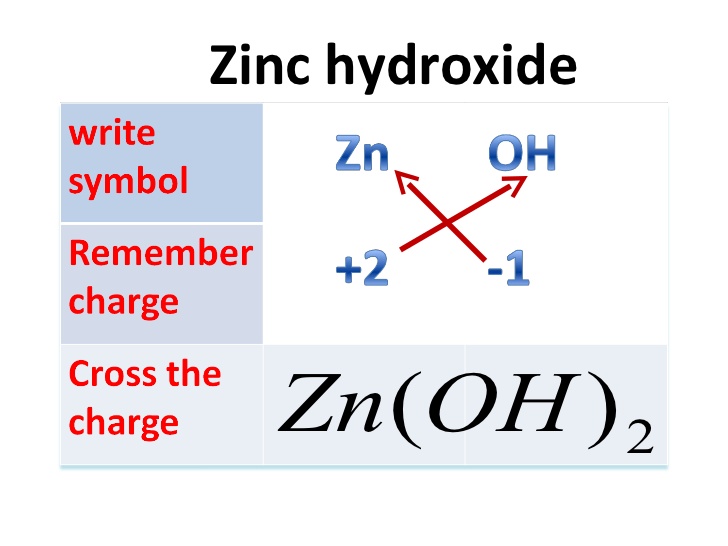
Task 5) Recall how to determine the Chemical Formula of Ionic Compounds
Ionic compounds are when positive and negative ions chemically bond together. This usually occurs between metal ions and non-metal ions. An example is the salt, sodium chloride or NaCl. We are mainly going to look at ionic compounds which are formed by positive and negative ions coming together.
|
Complete
* Read section 5.6) Ionic Compounds * Complete questions from the Activities Q8, 9, 10 and 11 * Complete worksheet 4.2) Naming and Writing Chemical Formulas. If you are experiencing any difficulty, watch the video below *Complete worksheet 2) Chemical Formulas |
Interesting Information - Ions in Action
|
The magnificant Southern Aurora which looks like amazing curtains of light in the sky are produced by ions streaming in from the Sun and trapped along the Earth's Magnetic Field. Read more about this beautiful natural phenomena on the Australian Geographic article "Aurora Australis" |
|
|
|
Extension Activity) Learn about Covalent Compounds
|
Extra Resources and Answers
1. Activity 3.1) Ionic Compounds in Science Focus 3 Homework Book. To learn about chemical formulas and the naming of ionic compounds 2. Activity 1.1) Writing Formulas in Science Focus 4 Homework Book. To further learn about chemical formulas and the naming of ionic and covalent compounds. 3. View Video Chemical Bonding and then answer questions in documents Ionic Bonds Notes and Covalent Bonds Notes. 4. Answers to Worksheet 4.2) Naming and Writing Chemical Formulas 5. Answers to worksheet 2) Chemical Formulas |
|
|
2) Information on Physical and Chemical Changes
Pre-test - See what you remember about Physical and Chemical Changes. Provided Notes on Physical and Chemical Changes. Read CS4 pg 372 - 374 to further develop your understanding. Practice Questions - SF3 Unit 3.1) Questions pg 71 Q1 - 17 Practical task |