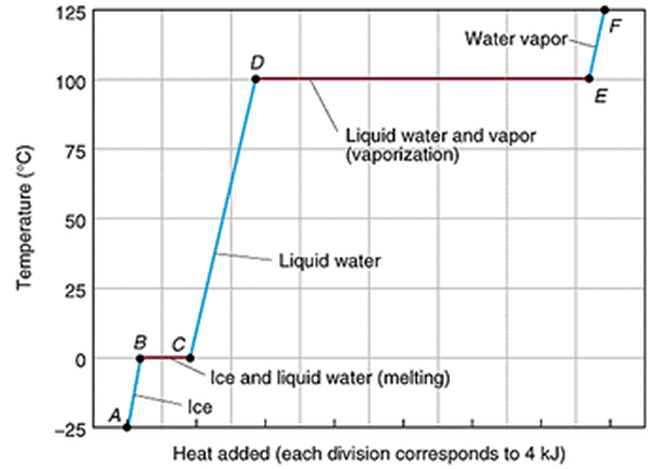
1.6) Changes of State
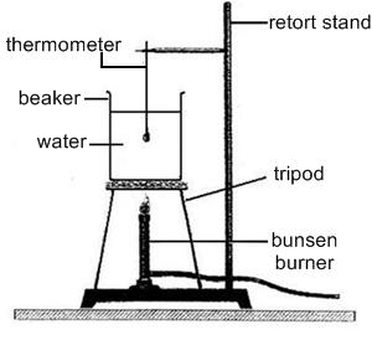
Activity : Where does the ENERGY go???
Aim: To investigate what happens to the temperature of water as it changes state
Method:
1) Mix 3 - 6 ice cubes with 100 mL of water in a 250 mL beaker.
2) Set up experiment as shown and record the initial temperature
3) Put the Bunsen Burner on the blue flame and record the temperature every minute until the water has boiled for 3 minutes
Risk Assessment:
Results;
1) Create a table with the correct headings
2) Draw a graph of temperature on the vertical axis and time on the horizontal axis
Questions:
1) Explain why you had to keep the airhole open during the experiment.
2) Your graph probably didn't start at 0 degrees C. Explain why.
3) Identify any level sections in your graph. Explain why these sections occur.
4) Imagine you were able to measure the temperature of the steam when the water is boiling for 5 minutes as you continued to heat it. Describe what you think would happen to the temperature? Would it increase, decrease or stay at 100oC?
Aim: To investigate what happens to the temperature of water as it changes state
Method:
1) Mix 3 - 6 ice cubes with 100 mL of water in a 250 mL beaker.
2) Set up experiment as shown and record the initial temperature
3) Put the Bunsen Burner on the blue flame and record the temperature every minute until the water has boiled for 3 minutes
Risk Assessment:
Results;
1) Create a table with the correct headings
2) Draw a graph of temperature on the vertical axis and time on the horizontal axis
Questions:
1) Explain why you had to keep the airhole open during the experiment.
2) Your graph probably didn't start at 0 degrees C. Explain why.
3) Identify any level sections in your graph. Explain why these sections occur.
4) Imagine you were able to measure the temperature of the steam when the water is boiling for 5 minutes as you continued to heat it. Describe what you think would happen to the temperature? Would it increase, decrease or stay at 100oC?
|
Video: Changing States of Matter
(30 mins) |
| ||||||