IQ 1) What are ATOMS made up of?
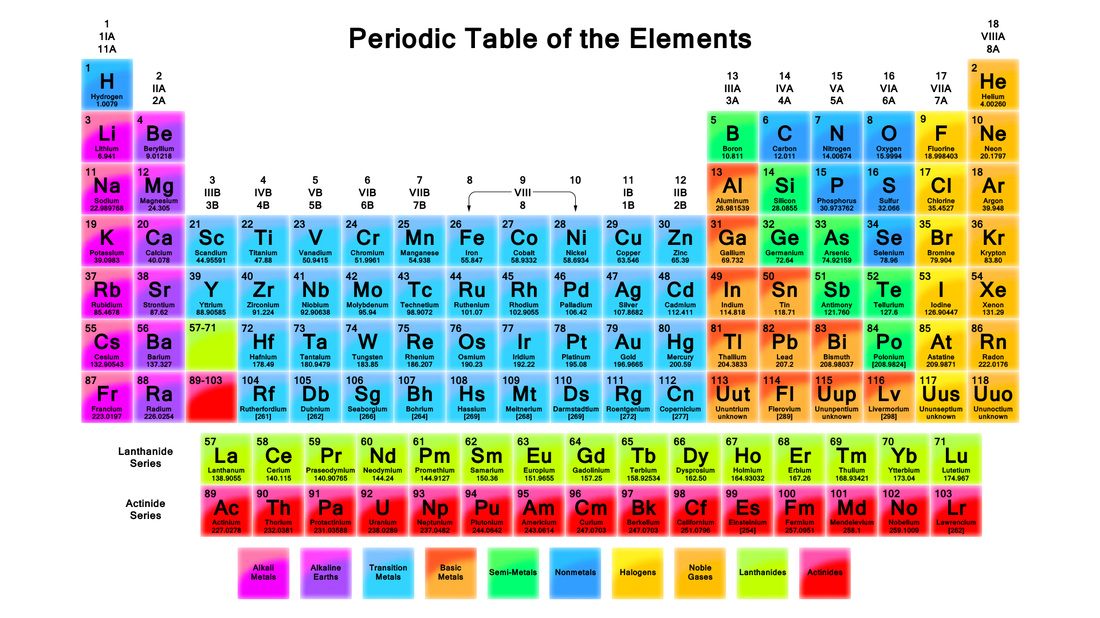
Around 1860, there was over 60 known elements and scientists were studying their properties and trying to see patterns in their properties so that they could be arranged in a table. In 1869, a Russian scientist Dmitri Mendeleev worked out a system for sorting elements into groups. It was called the Periodic Table and is still used today however there are now 118 known elements.
So how are these elements grouped??
1) Firstly each element is given a number that represents the number of protons (positively charged particles) that are found in the atom.
2) Then each element was placed in groups that have similar properties. The groups are METALS, METALLOIDS, SEMI-METALS, NON-METALS and NOBLE GASES. To identify these groups, colour your periodic table as indicated below which is slightly different to the image of the periodic table given:
Metals - Purple
Metalloids - Light Blue
Semi-Metals - Light Green
Non-Metals - Red
Noble Gases - Yellow
So what does an atom look like?
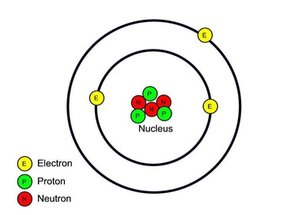
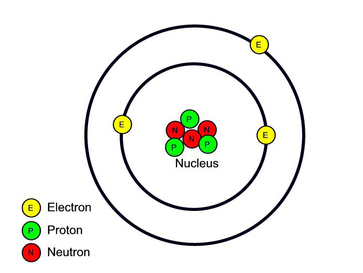
- * Atoms consist of PROTONS (p), NEUTRONS (n) and ELECTRONS (e).
- * Protons and Neutrons are found in the NUCLEUS
- * Electrons orbit around the nucleus.
- * Protons are positively charged, neutrons are neutral and electrons are negatively charged.
To draw an atom, we need to work out the number of protons, neutrons and electrons using the Periodic Table.
|
The Number of Protons and the Number of Electrons:
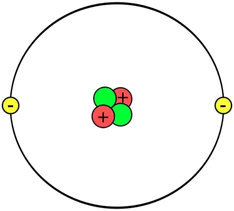
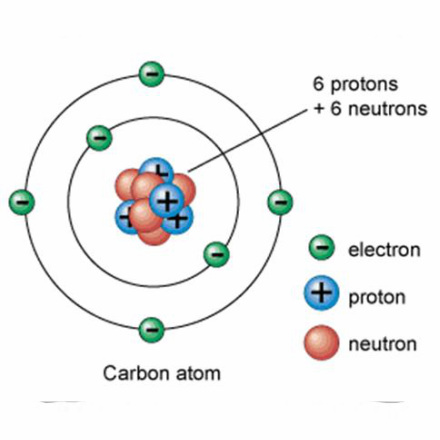
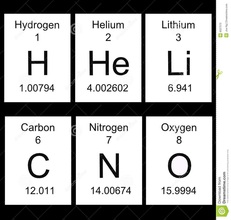
The number of protons is determined from the periodic table. It is the smaller of the two numbers given for an element. This number has a special name - The Atomic Number. The number of electrons for an atom is the same as the number of protons. This is the LITHIUM atom. It is the THIRD element on the Periodic Table and this means that it has THREE PROTONS. It's atomic number is also 3. It also has three electrons. |
|
The Number of Neutrons:
The number of neutrons is found by using both numbers given in the periodic table for that element. The larger number is called the ATOMIC MASS. The number of neutrons is found by Neutrons = Atomic Mass - Atomic Number Hence lithium will have 7 - 3 = 4 neutrons. Also carbon will have 12 - 6 = 6 neutrons |
|
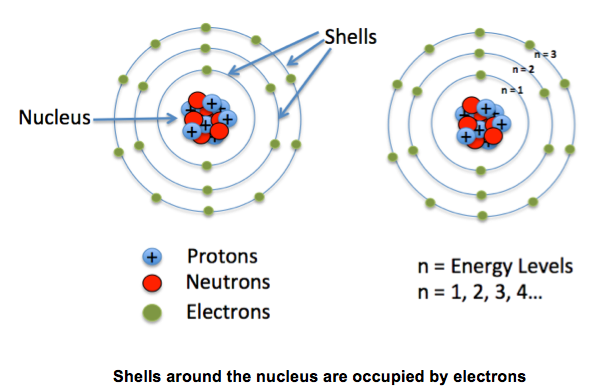
Electron Orbits
The Electrons are nearly 2000 times smaller than protons and neutrons. These electrons fly a long way away from around the nucleus in orbits. Sometimes the orbits are called shells or energy levels. They follow the rule 2n2 Orbit 1 = 2 electrons Orbit 2 = 8 electrons Orbit 3 = 8 electrons Orbit 4 = 8 electrons |
Homework
1) Worksheet 5.1) The structure of the atom
2) Worksheet 5.2) Electron Shells
2) Read Chem4Kids - Atoms are the Building Blocks
1) Worksheet 5.1) The structure of the atom
2) Worksheet 5.2) Electron Shells
2) Read Chem4Kids - Atoms are the Building Blocks
|
Ground State, Excited State
Task 1) Demonstration Practical: Fireworks Colours Aim: To Identify elements by the coloured flames they produce Results: Create a table with the column headings - solution, compound formula, colour of flame, Metallic Element in solution Task 2) Something very special is happening here but before we can learn what is happening we must understand the structure of atoms. Read and make notes from the website "What causes the colours in flames?" Task 3) How has our understanding of the excited state and ground state electrons helped us make fireworks? Read the website "Fireworks" to answer this question. |
|
Extension Task on Atomic Model:
1) Read through this interesting website called "The Particle Adventure". It will give you a better picture of the building blocks of matter. Look at the menu on the left side and start of with "What is Fundamental?" |
History of Atomic Theory - Group Assignment and Peer Marking
|
Some helpful resources 1) The youtube video by Crash Course Chemistry 2) The BBC - GCSE Bitesize website "Development of Atomic Theory" 3) Create Atoms with this Shell-Shocked Interactive |
|
|
Further Resources:
1) For more practise, you can read through this interactive on the Periodic Table and there is a quiz at the end to test your understanding of the Periodic Table and the Atom. 2) Also WebElements is an outstanding interactive periodic table that contains significant information about different elements. 3) More Information on how to draw atoms is found on "Drawing Atoms" 4) More about atoms is found on "Atoms- The Basics" |
5) Make your own Atom Model - v2
In pairs, construct a correct 3D atomic model of an element by following the steps below:
Important Criteria for your Atomic Model
In pairs, construct a correct 3D atomic model of an element by following the steps below:
- Select an Element. Make sure that it's atomic number is less than 30 otherwise your model may be too complex to create!
- You must use materials other than paper such as Styrofoam balls, wood, balloons, food, pipe cleaners etc.
- Separate colours must be used for each part of the atom.
- A colour key must accompany your model.
- Include detailed description about the element that you chose as well as its Periodic Square.
- Include interesting properties about the element
- Also include uses of this element in society
Important Criteria for your Atomic Model
- The model should have the proper number of protons, neutrons and electrons.
- The atomic model must be properly labeled
- Your model must be scientifically accurate and creative