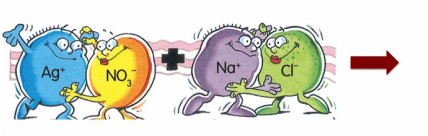
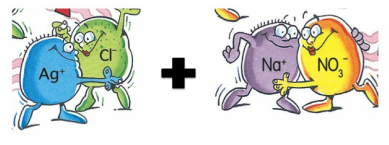
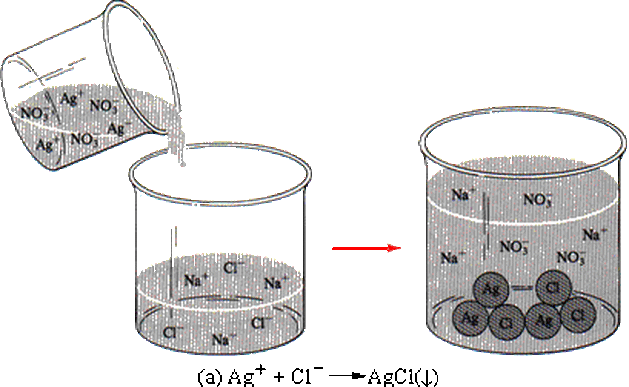
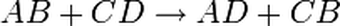4.4) PRECIPITATION REACTIONS
|
Task 1) Write all notes provided and make sure that you understand them
|
For example:
silver nitrate + sodium chloride ------> ??????
silver nitrate + sodium chloride ------> ??????
Task 2) Complete worksheet on writing Precipitation Reactions
To write an unbalanced chemical equation,
a) write the word equation
b) directly underneath the word equation, write the unbalanced chemical equation
c) Add subscripts – (s), (l), (g) or (aq). These symbols have the following meaning - (s) solid or precipitate, (l) liquid (only water), (g) gas and (aq) aqueous which means it is a powder mixed with water. For precipitation reactions, all the substances will be aqueous except ONE of the PRODUCTS.
i) Download worksheet "Constructing Word Equations" and complete steps (a) - (c) for each reaction.You can watch the youtube videos below if you need further clarification. Answers to the worksheet can be found
To write an unbalanced chemical equation,
a) write the word equation
b) directly underneath the word equation, write the unbalanced chemical equation
c) Add subscripts – (s), (l), (g) or (aq). These symbols have the following meaning - (s) solid or precipitate, (l) liquid (only water), (g) gas and (aq) aqueous which means it is a powder mixed with water. For precipitation reactions, all the substances will be aqueous except ONE of the PRODUCTS.
i) Download worksheet "Constructing Word Equations" and complete steps (a) - (c) for each reaction.You can watch the youtube videos below if you need further clarification. Answers to the worksheet can be found
|
|
|
|
Practical "Precipitation Reactions"
1) Conduct this fun practical to complete the table provided 2) If you are not confident, you can click here to check that you have correctly identified the precipitates. Extension work - If you are feeling confident see if you can read the table below correctly and check your answers to the practical 3) Complete the Constructing Word Equations and Chemical Equations Worksheet - This is a harder sheet that contains all different types of reactions - not just precipitation reactions. Download the sheet and you will notice that the reaction is written in words. Turn these words into word equations and then turn the words into chemical equations. If you are feeling confident you can balance these equations as well. However balancing is not necessary. Answers |
Extension Activity 1: Balancing Chemical Equations
Read Section 7.2) The Language of Chemical Reactions from Core Science Stage 5 textbook. An Additional Rule that is VERY VERY helpful is to balance your atoms in the following order - Metals, then Non Metals, then Oxygen and finally Hydrogen. You can also watch the youtube clips provided.
Also there is a very good website "Balancing Chemical Equations" which has tutorials, worksheets and quizzes to complete.
Read Section 7.2) The Language of Chemical Reactions from Core Science Stage 5 textbook. An Additional Rule that is VERY VERY helpful is to balance your atoms in the following order - Metals, then Non Metals, then Oxygen and finally Hydrogen. You can also watch the youtube clips provided.
Also there is a very good website "Balancing Chemical Equations" which has tutorials, worksheets and quizzes to complete.
|
|
|
Resources
| 1.6_precipitation_experiment_-_answers.pdf | |
| File Size: | 177 kb |
| File Type: | |