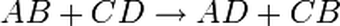|
Essential Question Why doesn't MATTER last FOREVER? 1.11) So..... Why doesn't MATTER last forever? 1.12) Reaction Type 1 - Precipitation Reactions 1.13) Reaction Type 2 - Corrosion 1.14) Reaction Type 3 - Combustion and Respiration 1.15) Rates of Reaction - Temperature, Surface Area and Catalysts 1.16) New Materials |
The Core Learning Intentions are: 1. Write word equations for common reactions 2. Describe some common reactions including: a) Precipitation b) Corrosion c) Combustion and Respiration d) Decomposition (review from year 9) 3. Understand that a chemical reaction is simply the rearrangement of atoms. 4. Describe the Law of Conservation of Matter when explaining chemical reactions. 5. Describe the difference between endothermic and exothermic reactions 6. Describe factors that affect the rate of reaction - temperature, surface area and catalysts. |
1.11) So..... Why doesn't MATTER last forever???
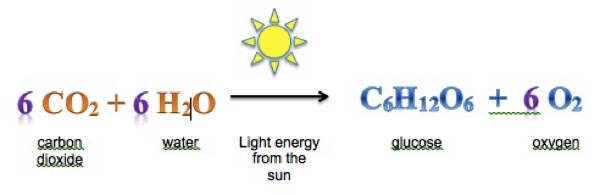
Chemical Reactions are occurring all the time and all around you. Useful materials such as cloth, paper, ink, ceramics, metals and polymers are all products of chemical reactions. Chemical reactions occur in the cooking and digestion of your food, and a very special chemical reaction, respiration, must occur in all the cells of your body to sustain life. Medicines help cure illness by affecting the chemical reactions in your body. Even everyday occurrences — the rusting of an old car, the burning fire of the barbeque and the fizzing of a bath bomb — are the result of chemical compounds interacting.
Chemical Reactions are when new substances are made by rearranging atoms. Think of it as the atoms are changing partners to create new substances. Hence MATTER doesn't last forever but the ATOMS that make up the matter can last forever!!!!
Chemical Reactions are occurring all the time and all around you. Useful materials such as cloth, paper, ink, ceramics, metals and polymers are all products of chemical reactions. Chemical reactions occur in the cooking and digestion of your food, and a very special chemical reaction, respiration, must occur in all the cells of your body to sustain life. Medicines help cure illness by affecting the chemical reactions in your body. Even everyday occurrences — the rusting of an old car, the burning fire of the barbeque and the fizzing of a bath bomb — are the result of chemical compounds interacting.
Chemical Reactions are when new substances are made by rearranging atoms. Think of it as the atoms are changing partners to create new substances. Hence MATTER doesn't last forever but the ATOMS that make up the matter can last forever!!!!
|
|
The Law of Conservation of Mass
Notice that the same atoms are present in both the reactants and the products; that is, no new atoms have been introduced during the reaction. The atoms that were present at the beginning of the reaction are still the same ones present at the end. The reaction has simply been a rearrangement of the atoms. This means that mass has not changed and hence the Law of Conservation of Mass has been supported. This Law states that mass cannot be created or destroyed during a chemical reaction. Complete the practical: Conserve that Mass! |
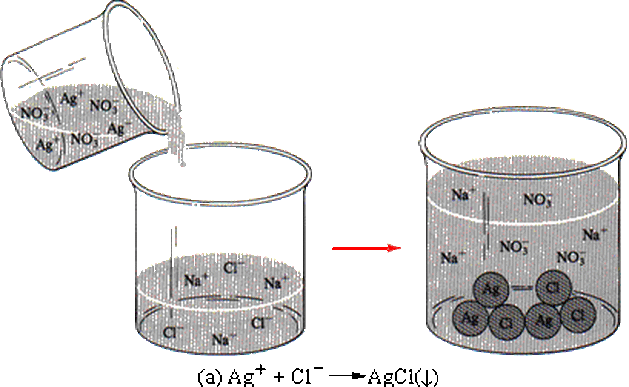
1.12) Reaction Type 1 - PRECIPITATION REACTIONS
For example:
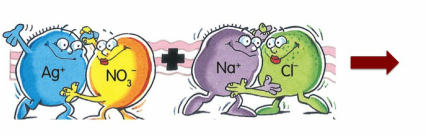
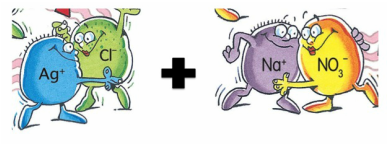
silver nitrate + sodium chloride ------> ??????
silver nitrate + sodium chloride ------> ??????
To write an unbalanced chemical equation,
a) write the word equation
b) directly underneath the word equation, write the unbalanced chemical equation
c) Add subscripts – (s), (l), (g) or (aq). These symbols have the following meaning - (s) solid or precipitate, (l) liquid (only water), (g) gas and (aq) aqueous which means it is a powder mixed with water. For precipitation reactions, all the substances will be aqueous except ONE of the PRODUCTS. You will need to use the solubility table to determine which PRODUCT is the reactant.
Another Example to write the Unbalanced Chemical Equation AND use Solubility Rules: see teachers board!!!!!
Written Task - Complete worksheet "Constructing Word Equations" to extend your knowledge of writing word equations. Check your answers. You can watch the youtube videos below if you need further clarification.
a) write the word equation
b) directly underneath the word equation, write the unbalanced chemical equation
c) Add subscripts – (s), (l), (g) or (aq). These symbols have the following meaning - (s) solid or precipitate, (l) liquid (only water), (g) gas and (aq) aqueous which means it is a powder mixed with water. For precipitation reactions, all the substances will be aqueous except ONE of the PRODUCTS. You will need to use the solubility table to determine which PRODUCT is the reactant.
Another Example to write the Unbalanced Chemical Equation AND use Solubility Rules: see teachers board!!!!!
Written Task - Complete worksheet "Constructing Word Equations" to extend your knowledge of writing word equations. Check your answers. You can watch the youtube videos below if you need further clarification.
|
|
|
|
Practical "Precipitation Reactions"
1) Conduct this fun practical to complete the table provided 2) Use the solubility table below to determine if there are any issues with your results. In other words are there reactions that shouldn't have produced a precipitate? 3) Complete the Precipitation Worksheet - Balance these equations using the information provided at the bottom of this page. |
1.13) Reaction Type 2 - Corrosion
|
You have probably seen the corrosion of iron on your bike, on pipes, nails etc. What is it and how can we stop it from occurring? Create a media item (video, pamphlet, newspaper article, TV Documentary etc) to create awareness about corrosion and its affect on our lives.
There is some information in SF4 on pages 17 - 19 otherwise find good websites to address the following questions: a) What type of reaction is corrosion? b) What is the general word equation for corrosion? c) How does the reactivity of the metal affect its ability to corrode? d) How does rusting occur? What factors are needed? e) What are some of the impacts of corrosion on society and the environment? f) Discuss two ways for protecting metals against corrosion. g) Outline how colorbond steel is constructed (ie the four layers). Identify the elements that are added to the steel. |
|
1.15) Rates oF Reaction - Temperature, Surface Area and Catalysts
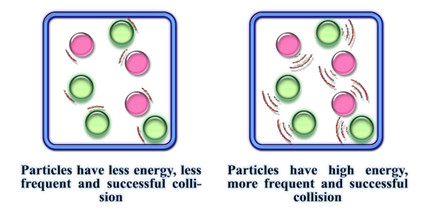
1) Conduct experiment "Effect of temperature on Reaction Rates". In your discussion, DESCRIBE how temperature affects the reaction rate.
EXTENSION: Use the information in the diagram to EXPLAIN how temperature affects reaction rate.
EXTENSION: Use the information in the diagram to EXPLAIN how temperature affects reaction rate.
|
2) Design an experiment to investigate how the surface area affects reaction rate. The details of the experiments are provided in SF4 pg 15 experiment 4) Rates of reactions 2. Your experiment must have the following headings:
a) Title b) Aim c) Equipment d) Method e) Results f) Discussion g) Conclusion Upload your report on Google Classroom |
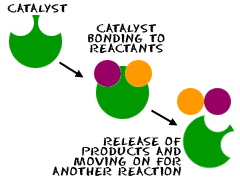
3) Conduct experiment 5) Reaction Rate - effect of Catalysts and Enzymes in SF4 pg 15. Write up experiment in your exercise book with a title, aim, results, questions and conclusion. In your conclusion, identify whether liver, manganese dioxide or apple/potato are catalysts of this reaction.
Then complete worksheet 1.4) Rates of Reaction in SF4 homework book
Then complete worksheet 1.4) Rates of Reaction in SF4 homework book
1.16) New Materials
Science not only seeks to understand how the world around us works but also attempts to use knowledge to make our lives better. One example is the development of new materials using our understanding of Chemistry.
New Materials
In pairs, choose a new material from the website above and prepare a short presentation to give to the class. You will need to include:
- where the product was developed (and by who)
- the technology that was used to develop the new material
- properties of the new material and explanation of how it works
- the purpose of the new material
- outline three ways that this new material can significantly affect people's lives
Extended Response Question - Analyse how social, ethical and environmental considerations can influence decisions about scientific research related to the development and production of new materials.
Science not only seeks to understand how the world around us works but also attempts to use knowledge to make our lives better. One example is the development of new materials using our understanding of Chemistry.
New Materials
In pairs, choose a new material from the website above and prepare a short presentation to give to the class. You will need to include:
- where the product was developed (and by who)
- the technology that was used to develop the new material
- properties of the new material and explanation of how it works
- the purpose of the new material
- outline three ways that this new material can significantly affect people's lives
Extended Response Question - Analyse how social, ethical and environmental considerations can influence decisions about scientific research related to the development and production of new materials.
Extension Activity 1: Balancing Chemical Equations
Read Section 7.2) The Language of Chemical Reactions from Core Science Stage 5 textbook. An Additional Rule that is VERY VERY helpful is to balance your atoms in the following order - Metals, then Non Metals, then Oxygen and finally Hydrogen. You can also watch the youtube clips provided.
Also there is a very good website "Balancing Chemical Equations" which has tutorials, worksheets and quizzes to complete.
Read Section 7.2) The Language of Chemical Reactions from Core Science Stage 5 textbook. An Additional Rule that is VERY VERY helpful is to balance your atoms in the following order - Metals, then Non Metals, then Oxygen and finally Hydrogen. You can also watch the youtube clips provided.
Also there is a very good website "Balancing Chemical Equations" which has tutorials, worksheets and quizzes to complete.
|
|
|
Resources
| 1.6_precipitation_experiment_-_answers.pdf | |
| File Size: | 177 kb |
| File Type: | |
| 25081202.pdf | |
| File Size: | 798 kb |
| File Type: | |